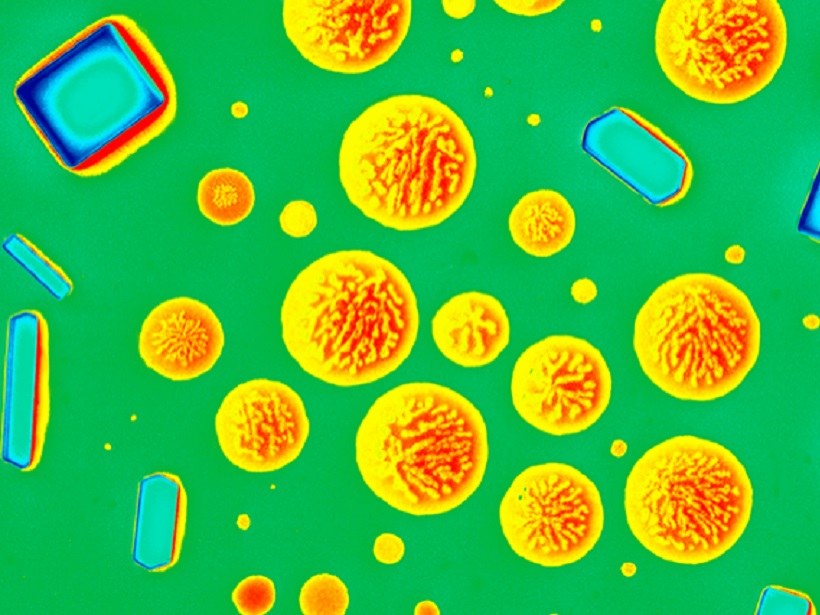
Aerosols are microscopic particles suspended in the atmosphere. Studying their characteristics and behavior is a rapidly developing area of atmospheric chemistry. A recent paper in Reviews of Geophysics focuses on aerosol mixing state, describing methods for measuring and modeling, as well as understanding the impacts on climate. Here, two of the authors of the paper give an overview of our understanding and recent developments in this scientific field.
What different sources of particles and atmospheric processes combine to form overall aerosol in the atmosphere?
There are countless sources for aerosols, but to keep it from being overwhelming, we can roughly categorize them as coming from combustion processes (soot, biomass burning, and industrial particles), from mechanical processes (mineral dust, sea salt, car brake dust), and forming directly from low-volatility gases.
The challenge we face is that particles are not simply emitted and remain the same through their lifetime.
The challenge we face is that particles are not simply emitted and remain the same through their atmospheric lifetime. Instead, various gases condense on or evaporate from the particles, and new species can form within the particles. Also, particles randomly collide with each other and form aggregates.
Thus, understanding the aerosol at any given time resembles hitting a moving target. The experimental methods and modeling approaches discussed in our review try to get a handle on this moving target.
What is ‘aerosol mixing state’ and what are the main factors that influence it?
At its simplest, aerosol mixing state is trying to say which chemical species are mixed with each other in individual particles and understanding how that contributes to the overall aerosol.
There are two extremes to mixing state, external and internal mixing. An external mixture means that all particles consist of just one chemical species, meaning a soot particle is just soot, a dust particle is just dust, etc. An internal mixture on the other hand means that every particle contains all the species in the overall aerosol in the same proportions (so a soot particle may also contain organic carbon, sulfate, nitrate, etc.).
The challenge is that it is very rare to have a perfectly externally mixed or internally mixed aerosol and usually we are somewhere in between. Aerosol mixing state is evolving in the atmosphere and a reflection of the processes that act on the particles during their lifetime.

How could you describe this in simpler language for a non-expert?
I like to think about my son’s plate at dinner, with each bite he takes as an aerosol particle. His plate starts as externally mixed, with green beans, chicken, squash and rice all separated on his plate, just like the aerosol particles as they enter the atmosphere from different sources.
Within a few minutes some of the green beans are in the squash, while some bites contain chicken and green beans. Then we add barbeque sauce (representing secondary species), which ends up on pretty much everything.
The bites are not an internal mixture, which would be the equivalent of the plate being put in a blender, with each bite having the same amount of everything. Thus, the plate and bites are each unique, but neither fully externally nor internally mixed. Various methods and models help us understand that complexity.
What techniques are used to measure aerosol mixing state?
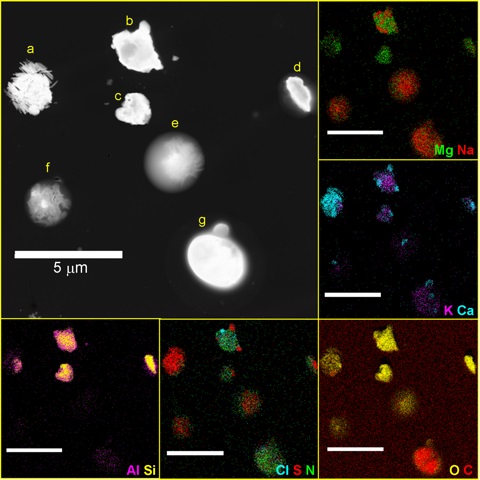
Any technique that can provide single-particle composition data can help us understand the mixing state of the atmospheric aerosol. The key is that a method has to be able to analyze in situ or collected samples particle-by-particle, so that the different distinct particle chemical compositions don’t get smeared together within an average.
The methods have been used the most to study aerosol mixing state each provide their own flavor of information: single particle mass spectrometry (molecules and fragments), electron microscopy with X-ray spectroscopy (elemental) and microspectroscopy (functional groups).
Researchers are constantly pushing to understand more about the physicochemical properties of individual particles, which are enabling us to probe even variations within particles in composition, surface properties, acidity, and more.
These advances are enabling us to learn more than ever about the mixing state of the overall aerosol.
What impact does aerosol mixing state have on the atmospheric properties that influence climate?
Aerosols influence climate because they impact the Earth’s radiative balance.
Aerosols influence climate because they impact the Earth’s radiative balance. This can happen directly as the particles scatter or absorb sunlight, or indirectly as the particles provide nuclei that help form cloud droplets and ice particles, thereby changing the reflectivity and lifetime of clouds.
The magnitude of these effects depends on the make-up of the individual particles and how they are assembled in a population.
For example, soot particles that are coated with organic material absorb light differently than a situation where soot and organics reside in separate particles. This changes the heating of the atmosphere where the aerosol resides. Similarly, the propensity to form cloud droplets or ice particles is different for coated soot particles compared to bare soot. These differences in the initial formation process of clouds propagate and determine how much sunlight clouds reflect back into space, a key process in determining the Earth’s energy budget.
What are some of the unresolved questions where additional research is needed?
Despite the very sophisticated measurement methods that are currently available, there is no single instrument that can capture aerosol mixing state in its entirety.
Despite the very sophisticated measurement methods that are currently available, there is no single instrument that can capture aerosol mixing state in its entirety. We therefore need to use multiple instruments that simultaneously sample the same aerosol and combine them in a way that yields a complete picture of aerosol mixing state. How to exactly do this, and how to connect these measurements to model output is challenging – but necessary for building a quantitative understanding of the aerosol mixing state in the ambient atmosphere.
Another important unresolved question is how to predict aerosol climate impacts – that is the aerosol impact on cloud properties and the aerosol interaction with light – from a known aerosol mixing state. The largest unknowns presently are associated with the formation of ice particles.
—Nicole Riemer (email: [email protected]), University of Illinois at Urbana‐Champaign; and Andrew Ault, University of Michigan
Citation:
Riemer, N.,Ault, A. (2019), The diversity and complexity of atmospheric aerosol, Eos, 100, https://doi.org/10.1029/2019EO124333. Published on 24 May 2019.
Text © 2019. The authors. CC BY-NC-ND 3.0
Except where otherwise noted, images are subject to copyright. Any reuse without express permission from the copyright owner is prohibited.