The ocean plays a major role in reducing the impact of climate change by absorbing both heat and carbon dioxide. To date it has taken up over 90 per cent of the excess heat in the climate system and about 25 per cent of the human released carbon dioxide. However, the uptake of carbon dioxide changes the chemistry of seawater, a process known as ocean acidification. This has major consequences for the organisms living in the ocean and reduces the role of the ocean as a carbon dioxide sink. A recent article published in Reviews of Geophysics explores ocean alkalinity and its impact on biogeochemical processes. Here, the authors give an overview of its role in carbon dioxide uptake and in limiting changes in ocean chemistry.
Why is carbon dioxide taken up by the ocean?
Some compounds gain protons and others lose protons when added to solutions. These are called bases and acids, respectively. When carbon dioxide enters seawater, it reacts with water to form carbonic acid, a weak acid. Carbonic acid dissociates partly into a proton and a bicarbonate ion: about 19 out of the 20 molecules of carbon dioxide entering the ocean are converted into bicarbonate.
Uptake and storage of anthropogenic carbon dioxide is related to this conversion of the gas carbon dioxide, that can exchange between the atmosphere and ocean, to the non-volatile ion bicarbonate. Alkalinity governs the efficiency at which this occurs and provides “buffering” capacity towards acidification.
What is alkalinity?
Alkalinity is “one of the most central but perhaps not the best understood concept in aquatic chemistry” [Morel and Hering, 1993]. Alkalinity is the excess of proton acceptors (bases) in a solution. The term alkalinity was already in use by chemists in the first half of the 19th century, but the alkalinity concept is still associated with confusion, because it is used in multiple disciplines each with their own definitions.
Freshwater scientists and geologists often use charge balance alkalinity, a quantifiable property that balances the excess of cations over anions. In the ocean, the bases of the carbon dioxide system (bicarbonate and carbonate) dominate alkalinity and oceanographers define alkalinity based on a titration of seawater with a strong acid. There are subtle differences between these two definitions that become important when studying biogeochemical processes such as primary production and calcification.
How to quantify ocean’s buffering capacity?
Alkalinity is considered a measure of a solution’s ability to resist pH changes: waters with high alkalinity have a high capacity to buffer against changes in proton concentrations, i.e. to reduce changes in pH. Quantification of ocean buffering capacity has led to the use of several indices by oceanographers, but unfortunately not always in a consistent way.
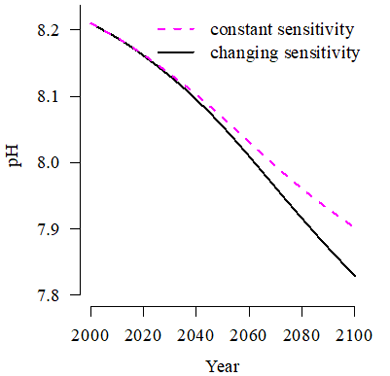
To prevent further confusion and propagation of inconsistencies, the term “sensitivity” should be added to factors that express the change in pH, or a related quantity, due to a change in concentrations, and the term “buffer factor” is best reserved for any quantity that expresses the resistance to change, i.e. the inverse of sensitivity. One well-known example is the Revelle factor, which expresses the change in partial pressure of carbon dioxide due to a change in the sum of dissolved carbon dioxide, bicarbonate and carbonate, and should be renamed the Revelle sensitivity factor.
What is the impact of biogeochemical processes on alkalinity?
Alkalinity is not only impacted by acid-base additions, but also by redox (reduction-oxidation) reactions and by mineral dissolution and precipitation.
Alkalinity is not only impacted by acid-base additions, but also by redox (reduction-oxidation) reactions and by mineral dissolution and precipitation.
Addition of a strong acid, such as sulphuric acid in acid rain, decreases alkalinity, while addition of lime increases alkalinity.
The breakdown of organic matter, an important example of an oxidation reaction, consumes alkalinity when oxygen is involved, while anaerobic degradation processes usually produce alkalinity.
Dissolution of minerals is often accompanied by alkalinity generation, while carbonate mineral formation consumes alkalinity. In fact, ecologists use changes in alkalinity to quantify calcification rates by corals and bivalves.
What governs whole ocean alkalinity levels?
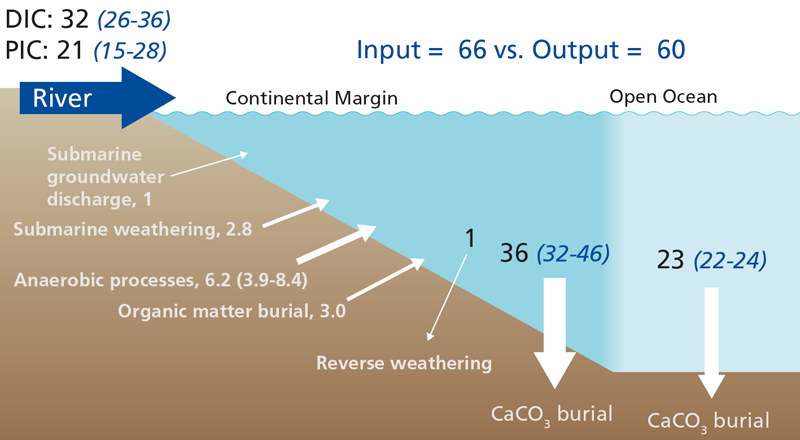
On geological time scales, whole ocean alkalinity is largely governed by the balance between ions generated by mineral weathering and removal of ions via the formation of calcium carbonate. Continental weathering on land generates alkalinity which eventually enters the ocean, where calcifiers produce skeletons and remove alkalinity.
Previous budgets concluded that the present-day ocean loses more alkalinity by carbonate burial than is supplied by rivers. Submarine groundwater inputs and within ocean generation of alkalinity by weathering and anaerobic breakdown of organic matter partly compensate for this imbalance, but, although it is poorly constrained, the particulate inorganic carbon flux to the ocean must also be included to close the alkalinity budget.
Why is it important to improve our understanding of alkalinity dynamics during times of global change?
The oceanic residence time of alkalinity is about 100,000 years, implying that changes will be moderate for decades and centuries to come. However, alkalinity measurements are highly accurate and reproducible.
The very small changes in alkalinity that can currently be detected serve as early warning signs for ocean acidification penetration into the ocean interior.
The very small changes in alkalinity that can currently be detected serve as early warning signs for ocean acidification penetration into the ocean interior. Sensitivity factors for the oceanic carbon dioxide system can be used not only to make projections for future pH or pCO2, but also to predict changes in seasonality, to attribute changes in ocean chemistry to external drivers or to evaluate the impact of ocean solutions such as alkalinisation on ocean chemistry and carbon dioxide uptake.
—Jack J. Middelburg ([email protected], ![]() 0000-0003-3601-9072), Earth Sciences, Utrecht University; Karline Soetaert, Royal Netherlands Institute for Sea Research (
0000-0003-3601-9072), Earth Sciences, Utrecht University; Karline Soetaert, Royal Netherlands Institute for Sea Research (![]() 0000-0003-4603-7100); and Mathilde Hagens (
0000-0003-4603-7100); and Mathilde Hagens (![]() 0000-0003-3980-1043), Wageningen University and Research
0000-0003-3980-1043), Wageningen University and Research
Citation:
Middelburg, J. J.,Soetaert, K., and Hagens, M. (2020), Understanding alkalinity to quantify ocean buffering, Eos, 101, https://doi.org/10.1029/2020EO146562. Published on 29 July 2020.
Text © 2020. The authors. CC BY-NC-ND 3.0
Except where otherwise noted, images are subject to copyright. Any reuse without express permission from the copyright owner is prohibited.

