Early life happily thrived in an oxygen-free world. These microbes, our earliest ancestors, subsisted on molecules such as hydrogen to make energy and lived in the hot inner recesses of Earth’s shallow crust. At some point, however, life developed a tolerance for oxygen, and evidence suggests it happened well before the world’s oceans and atmosphere were awash with the element.
New research shows that a stew of broken rocks, churned in near-boiling water, may have seeded this transition. The finding, published in Nature Communications, explains how life may have developed the biologic tools to deal with, and ultimately thrive in, oxygen-rich environments.
Rocks on early Earth had plenty of oxygen. But being locked in the solid crystal structures of minerals such as quartz and feldspar, it was mostly invisible to surrounding life. Only after the Great Oxidation Event around 2.4 billion years ago—when life learned how to photosynthesize—was “free” oxygen abundant in the oceans and atmosphere.
However, genetic reconstructions of 3.5-billion-year-old microbes suggest they possessed enzymes that could convert some of oxygen’s more reactive and damaging forms (such as hydrogen peroxide) into usable oxygen (O2) 1 billion years before the Great Oxidation Event.
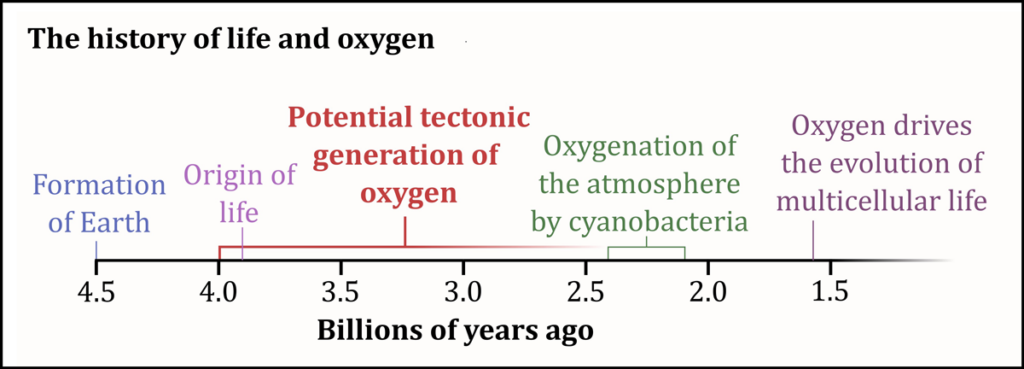
Until now, scientists have struggled to explain why microbes had the tools when the gas was virtually missing from the biosphere. Researchers have been exploring the idea of a nonbiologic source of O2 and reactive forms of oxygen to explain the mystery, said Tim Lyons, a biogeochemist at the University of California, Riverside, and head of the Alternative Earths Astrobiology Team.
Freeing Oxygen
While experimenting with chemical reactions that occur at the base of glaciers, where free oxygen is scarce, the researchers stumbled onto a way to generate hydrogen peroxide, said Jordan Stone, a doctoral student in geochemistry at Imperial College London and lead author of the study.
As part of his master’s project at Newcastle University, Stone filled thumb-sized glass tubes with either basalt or granite—analogues of oceanic and continental crust—sealed the tubes, replaced the oxygen with 100% nitrogen to simulate early Earth conditions, and then crushed the rocks. He then added water that had been stripped of free oxygen and heated the tubes. At temperatures above 80°C, hydrogen peroxide formed.
Similar conditions existed in Earth’s crust at mid-ocean ridges or at depths greater than about 1 kilometer. There, hot, oxygen-free, water-hosting early forms of life percolated. Fracturing the surrounding rocks—say, during an earthquake, when two sides of a fault crunch past one another—releases chemicals from imperfections in the rocks, which then react with the water to make hydrogen peroxide, according to the researchers. This reactive form of oxygen can break down in the environment, or microbes can convert it to O2, Stone said.
The process could have started when plate tectonics began, roughly 4 billion years ago, Stone said, giving microbes more than a billion years to adapt.
Early Life’s Oxygen Problem
“The role of free oxygen in life is a bit of a double-edged sword.”
“The role of free oxygen in life is a bit of a double-edged sword,” Lyons said.
Living in hot waters within Earth’s crust, the microbes would have been exposed to this geologically formed hydrogen peroxide. But this and other reactive forms of oxygen, produced as intermediate products of chemical reactions, are damaging to life, Lyons said. They destroy DNA and other life-sustaining molecules.
“Early life had to figure out how to deal with this,” Lyons said. “It was just an environmental hand that it was dealt.” In response, these early microbes evolved antioxidant enzymes that convert damaging reactive oxygen to something that is not harmful.
“[This period] kind of works as a stepping stone,” Stone said. As life evolved to protect itself from a low dose of hydrogen peroxide, it was developing the tools to thrive during the future onslaught of oxygen during the Great Oxygenation Event.
“Maybe if we can find out how life originated on Earth, then we can find out how life originated on other planets as well.”
“Later life developed this ability to take advantage of all the O2, through aerobic respiration,” Lyons said.
The work provides a realistic geologic explanation for early life’s antioxidant enzymes, Lyons said. “It’s a really nice study, and it addresses, I think, one of these really fundamental questions about life’s ability to adapt to things.”
An understanding of the complex interactions of life with its environment is helping scientists to answer quandaries beyond our world. “Maybe if we can find out how life originated on Earth, then we can find out how life originated on other planets as well,” Stone said.
—Jennifer Schmidt (@DrJenGEO), Science Writer
This news article is included in our ENGAGE resource for educators seeking science news for their classroom lessons. Browse all ENGAGE articles, and share with your fellow educators how you integrated the article into an activity in the comments section below.


