A translation of this article was made possible by a partnership with Planeteando. Una traducción de este artículo fue posible gracias a una asociación con Planeteando.
For decades, practitioners of radiocarbon dating have exploited a signal dubbed the “silver lining” of atomic weapons tests conducted in the 1950s. As blast debris mushroomed into the air, particles of radioactive carbon-14 entered the atmosphere. This fallout created the so-called bomb pulse: The atmospheric carbon-14 concentration spiked during the 1950s and early 1960s, followed by a gradual decline after the Limited Test Ban Treaty was signed in 1963.
The result is a distinctive blip in the carbon-14 curve, which has been a boon to the field of radiocarbon dating. But those methods were imperiled last year, when carbon dioxide (CO2) emissions from the combustion of fossil fuels undercut the precious signal. Consequently, researchers may have to rely on new or supplemental methods to date organic materials.
Carbon’s Atomic Clock
Radiocarbon dating exploits the fact that carbon occurs in several forms, of which carbon-12 is the most abundant. Far less common is the radioactive isotope carbon-14. Carbon-14 is produced when cosmic radiation collides with the atmosphere. The isotope then descends to Earth’s surface, where it is incorporated into plants and other organic matter. The moment an organism dies, a radioactive clock begins to tick: Carbon-14 atoms decay over time, reducing the concentration of radioactive carbon in the tissue. By measuring the amounts of both isotopes, scientists can determine a sample’s death date: The smaller the concentration of carbon-14 is, the older the sample is.
Radiocarbon dating can be applied to samples as old as 50,000 years. However, the method loses precision as one probes into the distant past, with results often suggesting several possible ages or including large uncertainties. The bomb pulse, on the other hand, enabled dating recent samples to within 1–2 years, an amazing degree of precision.
“While radiocarbon dating is commonly associated with archaeology and objects from further in the past, the bomb pulse was more relevant to a wide range of forensic scenarios,” explained Fiona Brock, a former radiocarbon chemist at Oxford University and a current member of the Cranfield Forensic Institute at Cranfield University, where she teaches and advises on radiocarbon dating. Researchers have used the bomb pulse to identify victims of the Korean War, expose art forgeries, and sniff out bogus wines and whiskeys.
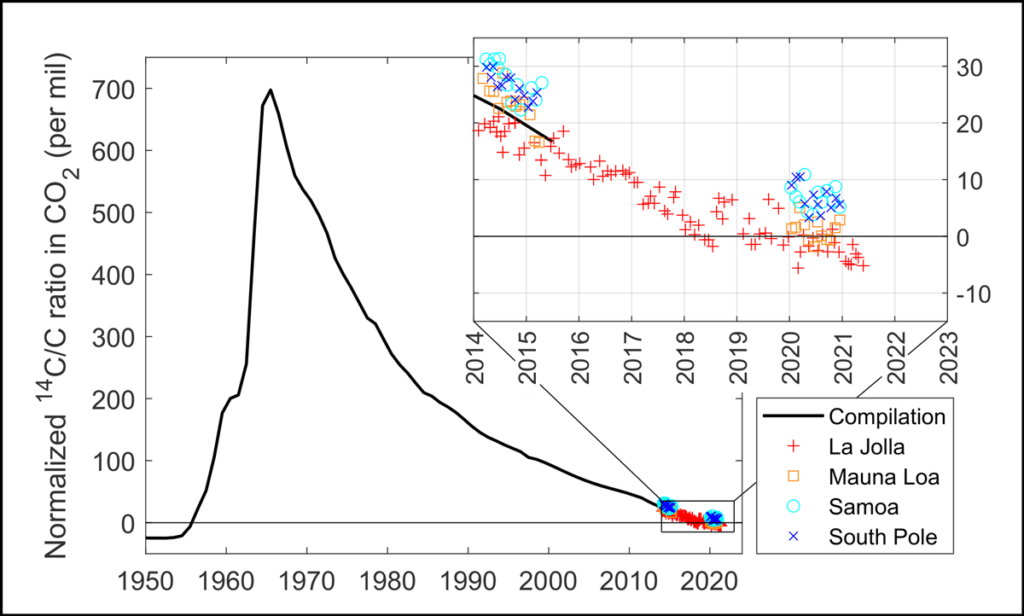
Radiocarbon Is Going Down
Ironically, this unexpected benefit of human meddling in the environment is falling victim to another kind of meddling: burning fossil fuels. Fossil fuels consist of organic material that is millions of years old—old enough that all its carbon-14 has decayed. Thus, the gases released during combustion of fossil fuels reduce the carbon-14 concentration in the atmosphere. Widespread use of fossil fuels is partially responsible for the rapid tapering of the bomb pulse after 1963.
Thirty years from today, radiocarbon dating will be unable to distinguish between a Viking tunic and a T-shirt fresh off the racks.
In 2021, the atmospheric concentration of carbon-14 dipped beneath prebomb values for the first time since the 1950s. This means organic tissue forming today has the same carbon-14 concentration as a sample from 1955—a problematic effect for researchers trying to distinguish samples with those ages. As we continue to burn fossil fuels, the problem will worsen: Thirty years from today, newly produced organic material will have the same carbon-14 concentration as a sample from 1050. This means radiocarbon dating will be unable to distinguish between a Viking tunic and a T-shirt fresh off the racks in 2050.
The loss of the bomb pulse affects both research and forensic applications. For instance, “good forgers could make the most of the situation where a modern paint has the same potential date as an historic artwork, or at least manipulate the situation to cause sufficient doubt as to whether something is genuine or a fake,” said Brock.
Rescuing Radiocarbon
The bomb pulse was doomed to fade out eventually as carbon-14 was incorporated into the ocean or decayed, but fossil fuel burning has hastened its demise. However, the loss of the bomb pulse doesn’t spell the end of radiocarbon dating. Other techniques can supplement radiocarbon data.
One such solution uses carbon-13, another stable isotope of carbon. Like its radioactive sibling, carbon-13 is scarce in fossil fuels, so its atmospheric concentration diminishes as we burn coal, oil, or gas. By measuring carbon-13 alongside carbon-14, researchers can determine whether a sample predates or postdates the Industrial Revolution. Alternatively, radioactive cesium-137 released during bomb tests can identify samples formed after 1963.
Peter Köhler, a physicist at the Alfred Wegener Institute in Germany who studies carbon isotopes and climate sensitivity, believes that radiocarbon dating will continue to be widely used. “One needs to apply common sense,” said Köhler. “Samples are measured within a context, and this should give enough information if there is a danger of mixing up modern and ancient.”
—Caroline Hasler (@carbonbasedcary), Science Writer
This news article is included in our ENGAGE resource for educators seeking science news for their classroom lessons. Browse all ENGAGE articles, and share with your fellow educators how you integrated the article into an activity in the comments section below.


