Carbon is one of the most important elements on our planet; its distribution on and in Earth affects the global climate system, the origin and evolution of life, and the types and availability of energy resources. The geological cycling of carbon, driven by plate tectonics over long timescales, is the main factor influencing the size of Earth’s shallow carbon reservoirs. Until recently, however, we had only a fragmented understanding of how much carbon resides in the deep Earth, its form, and how it moves between deep and shallow reservoirs.
Deep carbon science aims at understanding carbon in Earth’s crust, mantle, and core, where we now know more than 90% of Earth’s carbon resides.
Addressing these questions has been the core research goal of the Deep Carbon Observatory (DCO) program. Deep carbon science has emerged as a new scientific discipline, aimed at understanding the quantities, movements, forms, and origins of carbon in Earth’s crust, mantle, and core, where we now know more than 90% of Earth’s carbon resides. The program has amassed 7 years’ worth of discoveries about carbon in Earth’s depths. Over the next 3 years, the program will integrate these discoveries into an overarching model of carbon in Earth and create legacies for the scientific community and wider public.
DCO’s First 7 Years: A New Understanding of Deep Carbon
DCO research encompasses many related topics. How do subduction and volcanic and tectonic degassing cause carbon to cycle into and out of the mantle? What is the extent and diversity of the deep microbial biosphere? What forms and structures do carbon-bearing melts and minerals take in the mantle and core? What is the nature of deep sources of such carbon-based fluids as methane and higher hydrocarbons, and what processes control their formation [Hazen et al., 2013a]?
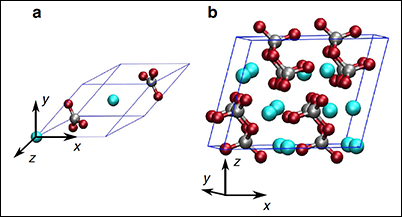
One highlight of our research is the discovery of what happens when carbon is carried from Earth’s crust into the mantle through subduction. For example, diamonds, which can contain geochemical signatures of organic material from Earth’s surface, may form as a result of pH shifts in mantle fluids [Sverjensky and Huang, 2015]. Deeply subducted carbonate minerals transform to a novel structure that features carbon in tetrahedral coordination with oxygen, rather than the triangular coordination more typical of minerals near the surface [Boulard et al., 2015] (Figure 1).
Closer to the surface, the geosphere and biosphere show a complex linked evolution. The diversity and ecology of carbon-bearing minerals on Earth have histories that closely mirror such major events in Earth’s history as the Great Oxidation Event, when biologically mediated free oxygen first appeared in our atmosphere, opening the way for the aerobic organisms we know today [Hazen et al., 2013b].
Recent research has extended the known limits to microbial life; one study showed that microbes thrive as deep as 2.5 kilometers in the oceanic crust [Inagaki et al., 2015]. Another study has identified unique microbial organisms that thrive as deep as 2 kilometers beneath the surface under the hot, highly saline conditions associated with the hydraulic fracturing of shale [Daly et al., 2016].

We’ve also improved our understanding of the volcanic output of carbon into our atmosphere: Novel instrument networks reveal that volcanic flux of carbon dioxide is twice what researchers previously thought [Burton et al., 2013] (Figure 2).

Other studies measure the fluxes of reduced carbon (e.g., methane) in diverse crustal environments. Next-generation mass spectrometry allows scientists to precisely identify the isotopic makeup of methane molecules (isotopologues) to trace them back to their sources in the crust and mantle [Young et al., 2016]. Still other studies have led to the discovery of “ancient water,” more than a billion years old (Figure 3). This discovery provides evidence for the existence of early crustal environments perhaps capable of harboring life [Holland et al., 2013].
Synthesizing a Decade of DCO Science
As the DCO completes its first decade of focused research, its emphasis is shifting toward scientific synthesis. For example, our workshops now center around unifying themes. Special publications and review articles will contain the underpinning science and discoveries that have emerged from the DCO and guide future scientific endeavors.
New models suggest that subducting slabs transport carbon more efficiently toward Earth’s surface than previously thought.
An important synthesis effort is the development of models and visualizations. We are developing models over a range of spatial and temporal scales to describe carbon mineralogy, carbon partitioning, fluxes between carbon reservoirs, and the extent and diversity of microbial life. Already, new models of the processes by which subduction causes Earth’s mantle to take in and release gas suggest that subducting slabs transport carbon more efficiently toward Earth’s surface than previously thought [Kelemen and Manning, 2015]. Scientists have attempted to extend such analyses back in time to the early Earth [Dasgupta, 2013].
Models of mantle melting beneath mid-ocean ridges [Rosenthal et al., 2015; Behn and Grove, 2015] show that small amounts of carbonated melts and their reactive flow play critical roles in mantle dynamics [Keller and Katz, 2016]. Another key modeling effort focuses on the nature of carbon-bearing fluids by integrating existing thermodynamic models of magmas in the crust (MELTS) [Ghiorso and Sack, 1995] and fluids in the mantle (Deep Earth Water, DEW) [Sverjensky et al., 2014a, 2014b] to form a framework for modeling mass transfer of carbon and other elements.
Reaching a Broader Audience
Deep Carbon Observatory scientists are striving to share what they have learned about the global deep carbon cycle with fellow scientists, future students, and an interested public.
DCO scientists are striving to share what they have learned about the global deep carbon cycle with fellow scientists, future students, and an interested public. We will share many of the findings through peer-reviewed publications, but DCO is also dedicating resources to creating films, infographics, and books. For more information, review DCO’s midterm report on deepcarbon.net and follow DCO on Twitter (@deepcarb) and Facebook.
Acknowledgments
DCO distributes seed funding from the Alfred P. Sloan Foundation via four research communities to leverage scientific endeavors addressing DCO’s goals. Robert Hazen, Craig Schiffries, Katie Pratt, and Darlene Trew Crist provided valuable input.
References
Aiuppa, A., et al. (2007), Forecasting Etna eruptions by real-time observation of volcanic gas composition, Geology, 35(12), 1115–1118, https://doi.org/10.1130/G24149A.1.
Behn, M. D., and T. L. Grove (2015), Melting systematics in mid‐ocean ridge basalts: Application of a plagioclase‐spinel melting model to global variations in major element chemistry and crustal thickness, J. Geophys. Res. Solid Earth, 120(7), 4863–4886, https://doi.org/10.1002/2015JB011885.
Boulard, E., et al. (2015), Tetrahedrally coordinated carbonates in Earth’s lower mantle, Nat. Commun., 6, 6311, https://doi.org/10.1038/ncomms7311.
Burton, M. R., G. M. Sawyer, and D. Granieri (2013), Deep carbon emissions from volcanoes, Rev. Mineral. Geochem, 75(1), 323–354, https://doi.org/10.2138/rmg.2013.75.11.
Daly, R. A., et al. (2016), Microbial metabolisms in a 2.5-km-deep ecosystem created by hydraulic fracturing in shales, Nat. Microbiol., 1, 16146, https://doi.org/10.1038/nmicrobiol.2016.146.
Dasgupta, R. (2013), Ingassing, storage, and outgassing of terrestrial carbon through geologic time, Rev. Mineral. Geochem., 75, 183–229, https://doi.org/10.2138/rmg.2013.75.7.
Ghiorso, M., and R. Sack (1995), Chemical mass transfer in magmatic processes IV. A revised and internally consistent thermodynamic model for the interpolation and extrapolation of liquid-solid equilibria in magmatic systems at elevated temperatures and pressures, Contrib. Mineral. Petrol., 119(2–3), 197–212, https://doi.org/10.1007/BF00307281.
Hazen, R., A. Jones, and J. Baross (Eds.) (2013a), Carbon in Earth, Rev. Mineral. Geochem., 75.
Hazen, R. M., et al. (2013b), Carbon mineral evolution, Rev. Mineral. Geochem., 75, 79–107, https://doi.org/10.2138/rmg.2013.75.4.
Holland, G., et al. (2013), Deep fracture fluids isolated in the crust since the Precambrian era, Nature, 497(7449), 357–360, https://doi.org/10.1038/nature12127.
Inagaki, F., et al. (2015), Exploring deep microbial life in coal-bearing sediment down to ~2.5 km below the ocean floor, Science, 349(6246), 420–424, https://doi.org/10.1126/science.aaa6882.
Kelemen, P. B., and C. E. Manning (2015), Reevaluating carbon fluxes in subduction zones, what goes down, mostly comes up, Proc. Natl. Acad. Sci. U. S. A., 112(30), E3997–E4006, https://doi.org/10.1073/pnas.1507889112.
Keller, T., and R. F. Katz (2016), The role of volatiles in reactive melt transport in the asthenosphere, J. Petrol., 57(6), 1073–1108, https://doi.org/10.1093/petrology/egw030.
Rosenthal, A., E. Hauri, and M. Hirschmann (2015), Experimental determination of C, F, and H partitioning between mantle minerals and carbonated basalt, CO2/Ba and CO2/Nb systematics of partial melting, and the CO2 contents of basaltic source regions, Earth Planet. Sci. Lett., 412, 77–87, https://doi.org/10.1016/j.epsl.2014.11.044.
Sverjensky, D. A., and F. Huang (2015), Diamond formation due to a pH drop during fluid-rock interactions, Nat. Commun., 6, 8702, https://doi.org/10.1038/ncomms9702.
Sverjensky, D. A., V. Stagno, and F. Huang (2014a), Important role for organic carbon in subduction-zone fluids in the deep carbon cycle, Nat. Geosci., 7, 909–913, https://doi.org/10.1038/ngeo2291.
Sverjensky, D. A., B. Harrison, and D. Azzolini (2014b), Water in the deep Earth: The dielectric constant and the solubilities of quartz and corundum to 60 kb and 1200° C, Geochim. Cosmochim. Acta, 129, 125–145, https://doi.org/10.1016/j.gca.2013.12.019.
Young, E. D., et al. (2016), A large-radius high-mass-resolution multiple-collector isotope ratio mass spectrometer for analysis of rare isotopologues of O2, N2, CH4 and other gases, Int. J. Mass Spectrom., 401, 1–10, https://doi.org/10.1016/j.ijms.2016.01.006.
Author Information
Marie Edmonds (email: [email protected]), Earth Sciences Department, University of Cambridge, UK; and Craig Manning, Department of Earth and Space Sciences, University of California, Los Angeles
Citation:
Edmonds, M, C. Manning (2017), Synthesizing our understanding of Earth’s deep carbon., Eos, 98, https://doi.org/10.1029/2017EO067913. Published on 21 February 2017.
Text © 2017. The authors. CC BY 3.0
Except where otherwise noted, images are subject to copyright. Any reuse without express permission from the copyright owner is prohibited.

