Editors’ Vox is a blog from AGU’s Publications Department.
Per- and polyfluoroalkyl substances (PFASs) are human-made chemicals used widely in a multitude of different applications and commercial products. While useful, these chemicals are potentially toxic to human health, wildlife, and the environment, and do not easily break down in the environment.
A recent article in Reviews of Geophysics explores the basics of PFASs and how they are transported in the subsurface. Here, we asked some of the authors to give an overview of these chemicals, how scientists study them, and what challenges remain.
What are per- and polyfluoroalkyl substances (PFASs) and how are they used?
Per- and polyfluoroalkyl substances (PFASs) are a family of humanmade chemicals containing thousands of members. Structurally, they are aliphatic substances with a fully or partially fluorinated hydrophobic alkyl chains (CF2–) of varying length and a terminal hydrophilic headgroup. The fluoroalkyl moieties provide excellent attributes, including heat, stain, and grease-resistant properties. Therefore, PFASs have been globally used in a wide spectrum of applications since the early 1940s, including cookware (Teflon), clothing, food packaging materials, personal care products, and fire-fighting foams. Two commonly used PFASs are perfluorooctanoic acid (PFOA) and perfluorooctane sulfonate (PFOS).


Where have scientists found PFASs in the environment?
PFASs have been widely detected in human blood, wildlife, and the environment. The atmosphere, aquatic environments (e.g., snow, precipitation, groundwater, river, lake, and seawater), as well as soil and sediment are frequently reported as important reservoirs for PFASs. Generally, atmosphere, surface water, and soil are the first repositories for PFASs. Transport of PFASs in these environmental compartments lead to their wide dissemination in oceans, deeper soil, groundwater, and the Arctic. Due to their persistent and bioaccumulative properties, PFASs can be accumulated in plants, animals, and other biota, ultimately entering human bodies.
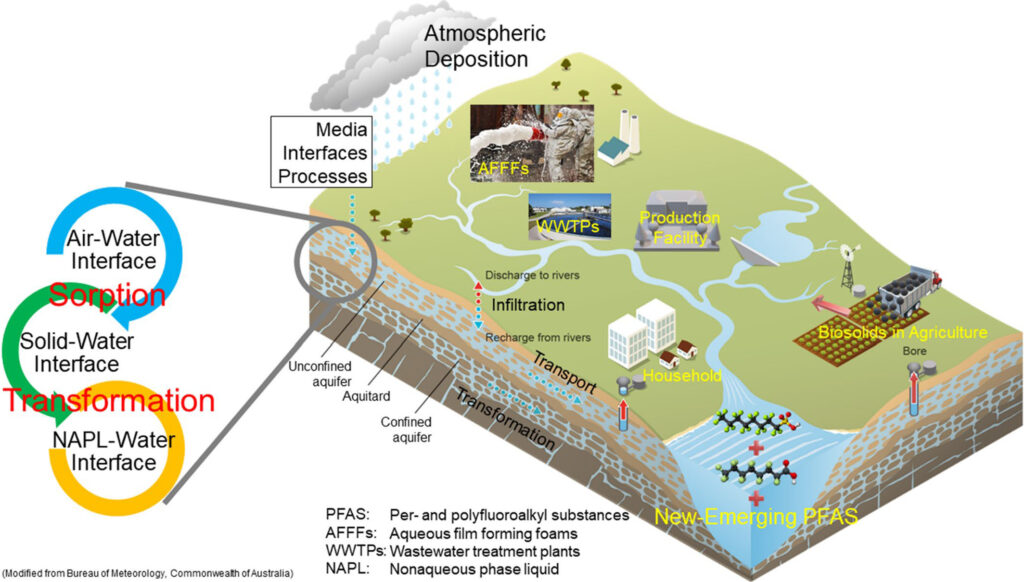
What dangers do PFASs pose to the health of humans and wildlife?
The potential health effects of PFASs on humans and wildlife have garnered growing concerns globally.
Some PFAS compounds have been listed as persistent organic pollutants (POPs) under the Stockholm Convention. The potential health effects of PFASs on humans (especially in susceptible populations, such as children and pregnant women) and wildlife have garnered growing concerns globally. Multiple lines of evidence have shown adverse impacts and risks on human health upon PFASs exposure. These include endocrine disruption, hepatic, reproductive, and developmental toxicity, kidney and testicular cancer, neurotoxicity, cardiovascular toxicity, immunotoxicity, and genotoxicity.
What methods do scientists use to detect and monitor PFASs?
A high-performance liquid chromatography (HPLC) connected to a tandem mass spectrometry system (MS/MS), or an ultrahigh-performance liquid chromatography (UPLC) coupled with a high-resolution mass spectrometry (HRMS) is normally used to analyze conventional PFASs. The HRMS, such as quadrupole time-of-flight mass spectrometry (QTOF-MS), can provide high resolving power, accurate mass, isotopic distribution, and MS/MS spectra. Therefore, it has been increasingly used to identify new PFAS compounds in the environment, such as cationic, zwitterionic, and neutral PFASs via non-targeted analysis.
What is the “vadose zone” and why is it important to chemical transport?
The vadose zone is the Earth’s terrestrial subsurface that extends from the surface to the regional groundwater table. Vadose zone includes surface soil, unsaturated subsurface materials, and a transiently inundated capillary fringe.
Surface soil of the vadose zone is an initial repository for PFAS release into the environment. The vadose zone can be a primary subsurface reservoir of PFASs, serving as long-term contaminant sources to the groundwater and surface water. Therefore, it is important to elucidate the chemical transport of PFASs in the vadose zone for risk assessment and designing and implementation of effective strategies for managing and treating PFAS contamination.
How have advances in modeling improved our understanding of PFAS transport?
Modeling can enable us to reliably assess the transport potential of PFASs in the vadose zone with complex conditions, especially their long-term transport at large scales.
PFAS transport in the vadose zone is a result of a combination of multiple retention mechanisms at multiple interfaces (e.g., sorption at the air–water interface and solid–water interface). Mathematical models can account for a comprehensive set of processes for PFAS transport under complex environmental conditions. Modeling can enable us to reliably assess the transport potential of PFASs in the vadose zone with complex conditions, especially their long-term transport at large scales.
What are some remaining challenges where additional research, data or modeling efforts are needed?
A large number of PFASs remain unidentified, or have been identified but not characterized well in terms of environmental occurrence, transport, transformation, and adverse impacts. Simple-to-use approaches for non-targeted or suspected screening of PFASs needs to be developed for identifying unknown PFASs. The transport and retardation of these new compounds in the subsurface soil and the aquifer remain largely unknown.
Future research should consider the spatiotemporal physical and geochemical heterogeneities of subsurface environments with a focus on model upscaling at different scales. Besides, sustainable and cost-effective techniques need to be further explored to efficiently manage and remediate PFAS contaminated sites.
—Dengjun Wang ([email protected]; ![]() 0000-0002-2047-5260), Auburn University, USA; and Xueyan Lyu, Nanjing University of Information Science and Technology, China
0000-0002-2047-5260), Auburn University, USA; and Xueyan Lyu, Nanjing University of Information Science and Technology, China
Editor’s Note: It is the policy of AGU Publications to invite the authors of articles published in Reviews of Geophysics to write a summary for Eos Editors’ Vox.

