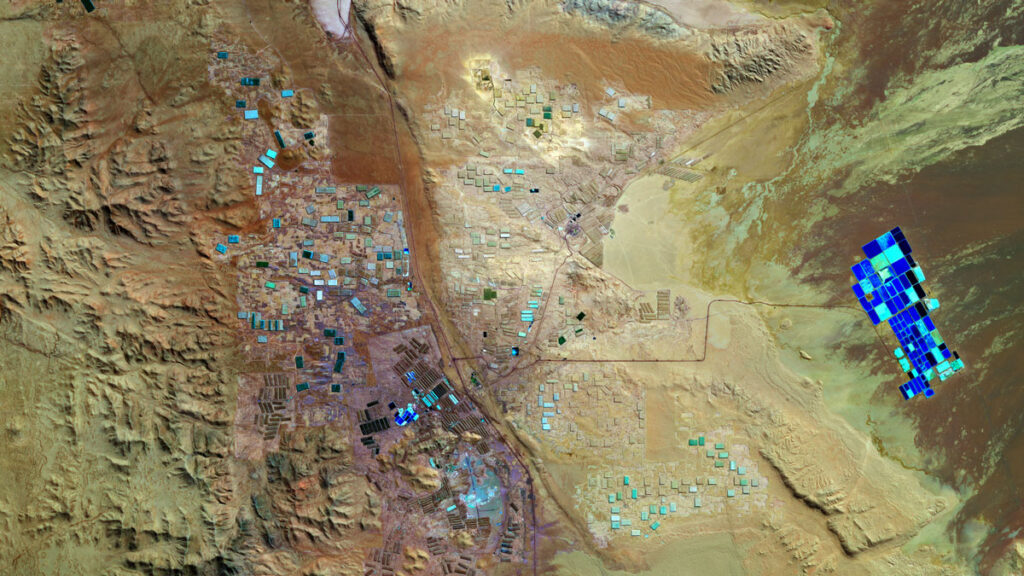
Lithium mines located in arid regions of South America, China, and the United States are striking when viewed from above, appearing as sprawling, colorful pools popping out from the desert like a giant painter’s palette. The open-air pools are filled with brine pumped from underground reservoirs. Once on the surface, the water eventually evaporates, leaving behind concentrated lithium. Today, much of the world’s lithium is extracted this way.
But what makes the mines so eye-catching is also a burden: Current mining methods using brine require vast swaths of land and water, and removing the brine from underground can cause freshwater reservoirs to flow into the open space, lowering the water table and contaminating water supplies in already dry regions. The evaporation process is also slow, taking 1–2 years.
Now, a new study published in Joule describes a novel lithium extraction method that is faster and potentially more environmentally friendly. The technique, which uses a unique chemical solvent, could also unlock lithium reserves in areas where conventional methods are infeasible because of land and water constraints. One such location is California’s Salton Sea, where brines contain enough lithium to build batteries for more than 370 million electric vehicles.
Flipping a Chemical Switch
The demand for batteries is driving scientists to develop more efficient lithium-extraction technologies, but the new study sprang from research on unique chemical solvents, called switchable solvents, that change properties under different conditions. Ngai Yin Yip, an environmental engineer at Columbia University and a senior author on the paper, was particularly interested in several switchable solvents that have an affinity for water at room temperature but repel it when it’s heated to 158°F (70°C).
“I like to think of this switchable solvent as a sponge.”
While studying this material, the team noticed that at room temperature, the solvent also attracted lithium, in addition to water. “I like to think of this switchable solvent as a sponge,” said Yip. “So it sponges up water and ions at lower concentrations.” Although the researchers don’t entirely understand why lithium interacts with the solvent this way, they think that the small size of lithium atoms might allow them to become encapsulated in water, essentially hitching a ride with the water.
The researchers started to see potential for lithium extraction and set up laboratory experiments to dig deeper. They mixed the solvent with beakers of brines, including one simulating the brine under the Salton Sea. When they mixed brines with the switchable solvent in laboratory beakers at room temperature, the solvent was attracted to water in the brine, and the solvent, water, and ions separated from the rest of the brine. The researchers then removed the layer of solvent, now containing water and lithium, from the rest of the brine, and raised the temperature. The heat switched the solvent to a hydrophobic state, in which it began “squeezing out” the water and ions for collection by the researchers, Yip said. The researchers then measured the amount of lithium and other positively charged metal ions, such as potassium, sodium, and magnesium, in the water.
While the water did contain small amounts of other cations, lithium was approximately 13 times more enriched in the solution than was sodium and 24 times more enriched than potassium in tests using the simulated Salton Sea brine.
Accessing the Inaccessible
“These kinds of technologies are really promising for having very low impact production of minerals.”
Yip said the new extraction method is much faster than current methods, and the solvents can also be reused to extract lithium from multiple batches of brine. The solvents are readily available and inexpensive, he said. “That was intentional, because we didn’t want to start off with a material that requires very elaborate synthesis.”
Alissa Kendall, a University of California, Davis industrial ecologist who was not involved in the study, found the study important because the Salton Sea region has geothermal power plants that could provide low-carbon heat, potentially even using waste heat from electricity generation. “These kinds of technologies are really promising for having very low impact production of minerals,” she said.
In future studies, Yip wants to better understand why the solvent pulls lithium along with water, as this interaction could be refined to improve the efficiency of lithium extraction. He also hopes to test the process’s scalability to determine whether industrial applications are feasible.
“This is really an engineered process,” Yip said, in that scientists both create the material and design the method to optimize its performance. “That can be helpful in terms of rapidly scaling up production to meet the forecasted increases in lithium demand over the coming decades.”
—Andrew Chapman (@andrewgchapman.bsky.social), Science Writer