Editors’ Vox is a blog from AGU’s Publications Department.
Carbon mineralization is a developing environment-focused technology that aims to minimize the increasing levels of human-made CO2 in the atmosphere to mitigate climate change. This approach works by injecting CO2 into underground rock masses, where chemical reactions convert the CO2 to mineral form, resulting in permanent storage.
A new article in Reviews of Geophysics explores recent developments from field, laboratory, and modeling studies of carbon mineralization. Here, we asked some of the authors to give an overview of carbon mineralization, insights from recent studies, and what questions remain.
How much could carbon mineralization potentially contribute to reducing atmospheric carbon dioxide and mitigating the global climate crisis?
A recent study suggests that the injection of CO2 into basaltic rocks alone could provide permanent storage capacity in the range of hundreds to thousands of gigatons of CO2 globally. Basaltic formations are abundant and widely distributed, making them a highly scalable option for mineral storage.
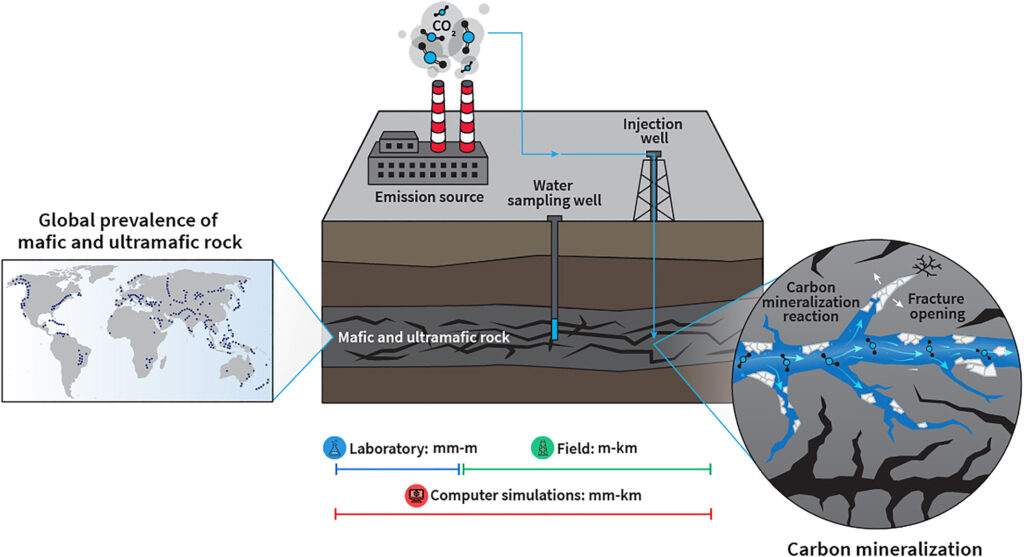
Why are mafic and ultramafic rocks more suitable for carbon mineralization than others?
Mafic and ultramafic rock masses are advantageous due to their prevalence in the Earth’s subsurface and their ability to rapidly store CO2 through carbon mineralization.
Mafic and ultramafic rock masses are advantageous due to their prevalence in the Earth’s subsurface and their ability to rapidly store CO2 through carbon mineralization. These rocks contain highly reactive silicate minerals abundant in metal cations (Mg2+, Ca2+, and Fe2+) that can react with dissolved CO2 to precipitate carbonate minerals, “locking” the carbon in the subsurface.
What role do fractures play in the process of carbon mineralization?
CO2 mineralization in ultramafic rocks, which tend to be more reactive but less permeable, has not yet been quantified. In these systems, fractures are expected to play a crucial role in the flow and reaction of CO2 within the rock mass and will influence the CO2 storage potential of the system. Therefore, their consideration is imperative to be able to predict CO2 mineralization at a specific storage site.
Where have field tests been carried out and what were the results?
Carbon mineralization in mafic and ultramafic rocks has been recently tested at the pilot scale. At CarbFix in Iceland, over 95% of the injected CO2 was mineralized in basalt after 2 years, while CarbFix2 mineralized >50% and 76% of the injected CO2 and sulfur, respectively, in four to nine months, and is currently still in operation. The Wallula Basalt Project in Washington State mineralized 60% of CO2 in basalt after 2 years, while the injection of CO2 into peridotite-rich Oman ophiolite is currently underway.
Field tests provide invaluable information about the physicochemical processes occurring in the subsurface during and after CO2 injection.
Field observations and data derived from these field tests provide invaluable information about the physicochemical processes occurring in the subsurface during and after CO2 injection, which can guide experimental designs and inform model parameters. The success of carbon mineralization field studies demonstrates the viability of storing CO2 in mafic and ultramafic rocks. However, reaching the levels of stored CO2 needed to reduce atmospheric CO2 requires a significant upscaling of the operations and the introduction of new field sites.
What insights have been derived from laboratory experiments?
Laboratory testing provides an opportunity to decipher the coupled chemical and mechanical processes that occur during CO2 injection and subsequent carbon mineralization in a controlled environment, which cannot be deciphered in complex field studies. An abundance of laboratory studies, including flow-through and batch-reactor experiments, have been carried out to understand the geochemical reactions that facilitate the carbonation of mafic and ultramafic rocks. These studies have produced data such as mineral dissolution and precipitation kinetics, mineralogy of reaction products, hydrodynamic parameters, and surface characteristics.
Fundamental research on the mechanical behavior of mafic and ultramafic rocks exists, and can be combined with studies on other materials (e.g., analog systems) that examine clogging and passivation of mineral surfaces, stress-corrosion cracking, and reaction-driven cracking to understand fluid flow and reactive transport in fractured rock.
How successful have models been at predicting fractures, flow, and reactions?
Models have been successful in providing insights on individual parts of the CO2 mineralization system. However, fully coupled studies of fracture, flow, geomechanics and geochemistry do not exist at the field scale. The coupled geochemical-geomechanical processes that affect fracture creation and damage, fluid flow, and mineralization in current models do not include all the relevant processes.
For mafic and ultramafic rocks, key questions such as stress-corrosion cracking and reaction-driven cracking require a fully coupled approach. These are key mechanisms that could result in positive feedback between fracture, reaction, and mechanics that allow for more and more reservoir rock to be accessed leading to a scalable approach for mineralization.
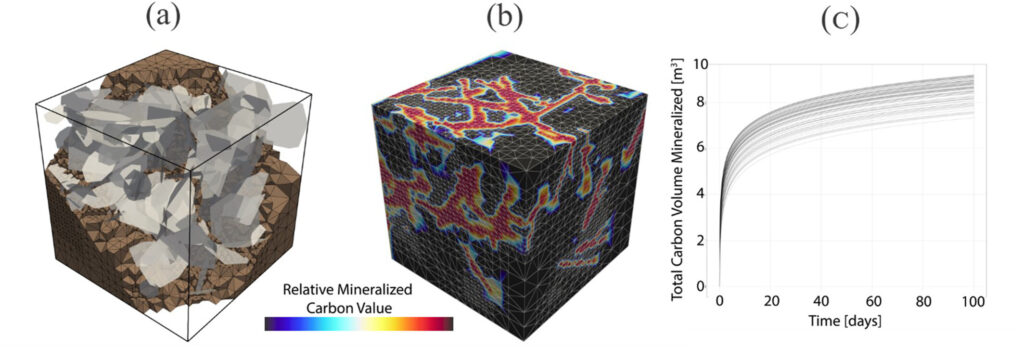
What are some of the challenges with the development of carbon mineralization models?
Geochemical and geomechanical models have developed separately over many decades. Therefore, coupling these models together with the added intricacy of dealing with complex fracture networks requires a new class of models.
What are some of the remaining questions where additional modeling, data, or research efforts are needed?
First, regarding the field scale studies of carbon mineralization, while rapid and highly efficient mineralization was observed in CarbFix, CarbFix2, and Wallula, generalization of these results to other potential field sites is necessary. Second, we need to refine the extensive published literature on experimental studies of carbon mineralization to gain insight into how this reaction will proceed within fractured systems.
Finally, the biggest perceived modeling challenge is coupling developed thermo-hydro-mechanical (THM) and thermo-hydro-chemical (THC) models to thermo-hydro-mechanical-chemical (THMC) models. THMC models can simulate the simultaneous dissolution and precipitation processes that drive carbon mineralization while also capturing how this process can drive fracturing through stress corrosion and reaction-driven fracturing processes.
—Haylea Nisbet ([email protected]; ![]() 0000-0002-8765-4677), Los Alamos National Laboratory, United States; and Hari Viswanathan ([email protected],
0000-0002-8765-4677), Los Alamos National Laboratory, United States; and Hari Viswanathan ([email protected], ![]() 0000-0002-1178-9647 ), Los Alamos National Laboratory, United States
0000-0002-1178-9647 ), Los Alamos National Laboratory, United States
Editor’s Note: It is the policy of AGU Publications to invite the authors of articles published in Reviews of Geophysics to write a summary for Eos Editors’ Vox.

